Pressure Is Read 75.5 Cm of Mercury
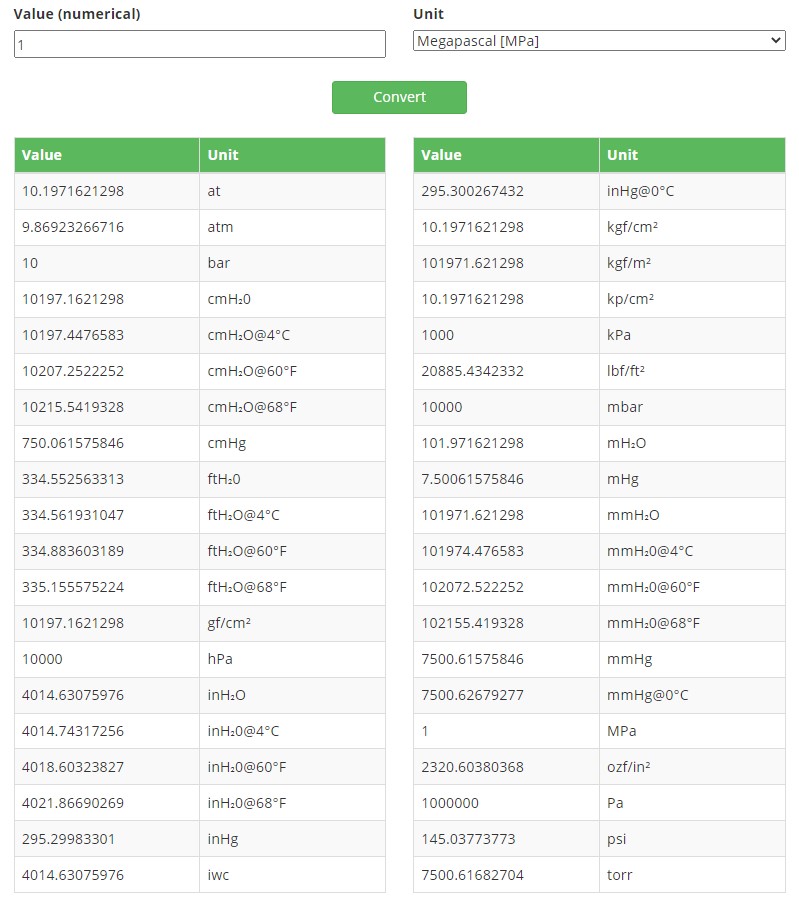
At that place are a lot of dissimilar pressure units in use around the world and sometimes this tin can be very confusing and may crusade unsafe misunderstandings. In this web log post, I will discuss the basics of dissimilar force per unit area units and different force per unit area unit of measurement families. If you just want to convert between different pressure level units, please check out our handy freepressure unit converter. At present that nosotros are talking about pressure, let's start with a video on how to calibrate a pressure transmitter: When I talk about force per unit area in this post, information technology does non refer to the stress you may be suffering in your piece of work, simply to the physical quantity. It is skillful to first accept a quick look at the definition of pressure, this volition also assist to ameliorate sympathize some of the force per unit area units. If y'all remember the studies of physics in school … as most of us don't remember… a short reminder is in order: pressure level is defined every bit strength per area perpendicular to the surface. That is often presented every bit formula p = F/A. The pressure is indicated with the letter "p", although the capital letter "P" can likewise exist seen being used on some occasions. And so what does this force per area hateful in practice? It means that in that location is a sure force affecting a specified area. When we look at forcefulness, it is specified as being Mass x Gravity. As there are so many different engineering units used for both mass and area, the number of combinations of these is huge. Plus there are also a lot of pressure level units that do not directly have the mass and expanse in their names, although it often is in their definition. It is good to notice that in do the "forcefulness" is non e'er included in the pressure unit names. For example, pressure unit kilogram-forcefulness per square centimeter should be indicated equally kgf/cm², but often information technology is indicated merely every bit kg/cm² without the "f ". Similarly, pound-strength per square inch (pfsi) is normally indicated every bit pounds per foursquare inch (psi). Download this article as a costless pdf past clicking the film beneath: Let'south beginning to look at the pressure units by looking at the SI system, which is the International System of Units, derived from the metric arrangement. Now that I mentioned the metric system, I tin already see some of yous taking a pace back… only please stay with me! SI arrangement is the world's most widely used organization of measurement. It was published in 1960 but has a very long history even before that. Pascal was named to honor the French physicist Blaise Pascal. Pascal (Pa) unit of pressure level is the basic pressure unit in the SI (the International System of Units) organisation. Pascal is non that one-time base unit of measurement is SI system, every bit it was adopted in 1971. Pascal is a and so-called "SI derived unit of measurement" of pressure level, equally it is derived from the base units specified in the SI system. The definition of ane Pascal is the pressure level of i Newton per square meter. And then, we can say one Newton is one kgm/south². To say Pascal (Pa) in a formula: Pascal is a very small pressure unit of measurement and is often used with different prefixes. Common multiples include hectopascal (1 hPa = 100 Pa), kilopascal (one kPa = 1 000 Pa) and megapascal (i MPa = 1 000 000 Pa). The standard atmospheric pressure is over one hundred thousand Pascals, to be verbal it is 101 325 Pa, or 101.325 kPa. In our (Beamex) accredited calibration laboratory we certificate all pressure calibrations in Pascals in calibration certificates (with suitable multiples). We unremarkably meet Pascal pressure unit being used in cleanroom differential pressure measurement transmitters or gauges, every bit the difference betwixt different rooms/areas is usually merely tens of Pascals. Out of Pascal's definition, the kg force can be replaced with different units like g (gram) force, and the meter can be replaced with centimeters or millimeters. Past doing that, we get many other combinations or pressure units, such every bit kgf/chiliad², gf/m², kgf/cm², gf/cm², kgf/mm², gf/mm², just to list a few. The unit "bar" is withal often used in some areas. It is based on the metric arrangement just is not role of SI system. Bar being 100 000 times Pascal (100 times kPa) it is anyway easy to catechumen. In some areas (like NIST in the USA) the bar is not recommended to exist used widely. And like for all pressure units, SI or not SI, we can use the common prefixes/coefficients in front end of them, near unremarkably used are milli (1/100), centi (1/10), hecto (100), kilo (k) and mega (1000000). To listing a few examples, that already gives us different Pa versions, all being normally used: Pa, kPa, hPa, MPa. The unit bar is most ordinarily used without prefix or with prefix milli: bar, mbar. Simply taking all mass units and combining those with all area units from the SI system, we get many combinations. Although the SI system is used in virtually countries, there are still a lot of other pressure level units also existence used. So let's have a look at those next. In countries using the Imperial system (like the USA and U.k.), the engineering units used both for mass and area are different than with the SI system. Therefore this also creates a whole new set of pressure units. Mass is beingness measured unremarkably in pounds or ounces, and area and distance with inches or anxiety. In the United states, the most common pressure unit of measurement is pounds per foursquare inch (psi). For process industries, a common unit is also inches of water (inH2O), which is derived from level measurement and the historical measurements of force per unit area differences with h2o in a column. The older pressure measurement devices were oftentimes made by using liquid in a transparent U-tube. If the pressure in both ends of the tube is the aforementioned, the liquid level on both sides is on the same level. Only if there is a divergence in the pressures, and so there is a difference in the liquid levels. The level deviation is linearly proportional to the pressure difference. In do, you lot can exit i side of the tube open up to the room's atmospheric pressure and connect the pressure to exist measured to the other side. As referred to the electric current atmospheric pressure, it is a gauge pressure type existence measured. The pressure scale is marked in the tube and then yous read the pressure by reading the difference in liquid levels. When pressure is applied it volition modify the liquid level and we can read the value. This sounds very elementary, no electronics and no wearing parts, so what could perhaps become wrong… well, let's see about that. The most commonly used liquid in the column was plainly water. But in order to be able to mensurate college pressure with a smaller U-tube, heavier liquids were needed. One such liquid is mercury (Hg) as it is much heavier than water (thirteen.6 times heavier). When you use the heavier liquid you don't need to have that long column to measure higher pressure, so y'all can make a smaller and more convenient size cavalcade. For example, blood pressure level was earlier (nonetheless sometimes is) measured with a mercury column. Mercury is mainly used because a water column for the same force per unit area range would exist so long it would not be applied to use it in a normal room, as the water column is nearly 13.vi times longer than the mercury column. Equally a result of this, even today the pressure unit of measurement that blood pressure is typically expressed is a millimeter of mercury (mmHg). A common industrial application for use of liquid column pressure units is to measure out the liquid level in a tank. For example, if you take a water tank that is xx feet (or 6 meters) loftier and yous want to measure the water level in that tank, it sounds pretty logical to install a force per unit area indicator with a calibration 0 to 20 feet of h2o, as that would tell directly what the water level is (13 feet in the instance film). Back to the water column: Information technology is clear that when the length indication was made to a U column, many different length units have been used, both metric and non-metric. This has generated many different pressure units. Although a liquid column sounds very uncomplicated, it is of import to remember that the weight of the liquid depends on the local gravity, and so if you lot calibrate the column in one place and have it to another (distant, different top) place, it may non be measuring correctly anymore. So gravity correction is needed to be precise. Besides, the temperature of the liquid affects the density of the liquid and that also affects slightly the readings of a U-tube. There are various different liquid column-based pressure level units bachelor, having the liquid temperature specified in the force per unit area unit of measurement, most commonly used temperatures are 0 °C, 4 °C, 60 °F, 68 °F. Only there are also water column units, which take no indication of the water temperature. These are based on a theoretical density of water, beingness 1 kg/1 liter (ISO31-three, BS350). In exercise, the water never has that high density. The highest density that water has is at +4 °C (39.2 °F) where it is approximately 0.999972 kg/liter. The density of h2o gets lower if the temperature is college or lower than +4 °C. Temperature tin have a pretty strong event on the density, for example going from +4 °C to +30 °C changes the water density by about 0.four%. Finally, the readability of a mechanical liquid column is typically pretty limited, and so you tin can't get very authentic measurements. And due to the mechanical limitations, you lot tin can't use a U-tube for high pressure. All of these above-mentioned bug make a U-tube liquid column not very practical to utilize. Besides, mod digital force per unit area measurement devices take replaced liquid columns. Just many of the pressure units created in the era of liquid columns have remained and are even so used today. To shortly summarize the liquid column-based force per unit area units: • For the length we have many units; mm, cm, m, inch and feet. By combining all of these, we go a long listing of pressure units, simply to mention a few: mmH₂O, cmH₂O, mH2O, mmHg, cmHg, mHg, iwc, inH2O, ftH2O, inHg, mmH2O@4°C, mmH2O@60°F, mmH2O@68°F, cmH2O@iv°C, cmH2O@60°F, cmH2O@68°F, inH2O@60°F, inH2O@68°F, inH2O@iv°C, ftH2O@60°F,ftH2O@68°F, ftH2O@4°C and then on. For measurement of the altercation' absolute pressure, dedicated pressure level units have been created. One of such is the standard atmosphere (atm) which is defined beingness 101325 Pascal. To add confusion, there is also a technical atmosphere (at) which is pretty shut, but not quite the same as atm. The technical atmosphere is ane-kilogram forcefulness per square centimeter. Then 1 at equals about 0.968 atm. Some other pressure unit of measurement used for measuring atmospheric absolute pressure is torr, existence i/760 of standard temper. And so torr is an absolute pressure unit, although that is typically not mentioned, you only need to know it, which can cause defoliation. Torr was initially meant to be the same every bit 1 millimeter of mercury, although the after definitions show a very pocket-sized difference in between. Torr is not part of the SI organisation. The abbreviation"cgs" comes from the words "centimetre-gram-second". As these words hint, the cgs system is a variation of the metric system, but instead of using the meter it uses centimeter as the unit of measurement for length, and instead of kilogram it uses gram as the unit for mass. The cgs is a pretty old system and has been mostly replaced kickoff by the MKS (meter-kilogram-second) system which and then has been replaced by the SI organization. Yet, you lot can notwithstanding sometimes run into cgs units of pressure. The cgs base of operations pressure unit isbarye (Ba), which equals 1 dyne per square centimeter. Dyne is the force needed to advance a mass of one gram to a rate of i centimeter per 2d per second. Every bit pressure level unit conversion, 1 barye (Ba) equals 0.1 Pascal (Pa). And some more… In addition to all the higher up pressure units, there are notwithstanding plenty Pressure unit of measurement conversions standards If you work with pressure level, you know that information technology is very common that pressure level is indicated with a certain pressure level unit and y'all need to convert it into another pressure unit. Pressure units are based on standards and the conversion between units should as well exist based on standards. The nearly common standards for pressure units are: Pressure unit converter tool I tried to brand a conversion table betwixt different force per unit area units, but that tabular array started rapidly to go a huge matrix that would not exist easy for you to utilize at all. So instead of making a conversion table, we developed an online pressure level unit of measurement converter for our website. With this converter, you can easily catechumen a pressure reading from one unit of measurement into other units. Please click on the link to bank check out thepressure unit converter. Below is an example screenshot from the pressure unit converter, when converting 1 MPa into other units: Interested in press this text or sharing it with your peers? Download this full article as a free White Paper from the movie link beneath: Click on this link to learn more about theBeamex pressure level calibration tools. Please cheque out our new electrical pressure pump Beamex ePG: 
It'south a jungle out there!
Table of contents
What is Pressure?

International System of Units (SI system) / Metric
Pascal unit, the SI unit of pressure
A Newton is a force needed to accelerate 1 kilogram of mass at the rate of one meter per 2d.
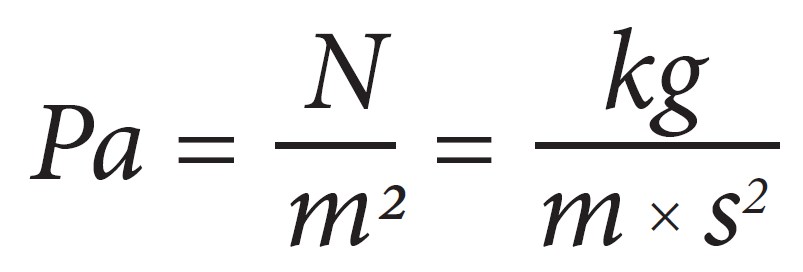
Calibrating these very low-pressure instruments is a challenge of its own.
Imperial units
Then some pressure units derived from these are lbf/ft², psi, ozf/in², iwc, inH2O, ftH2O. Liquid cavalcade units
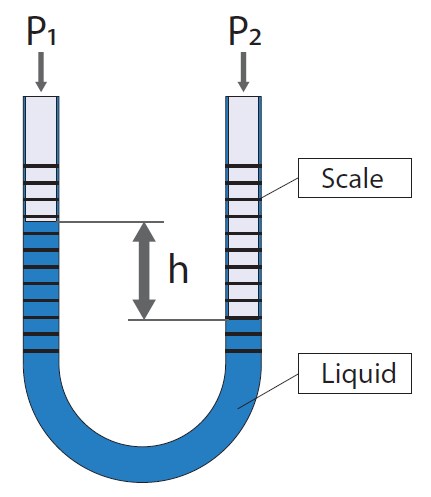

• So we have columns for dissimilar liquids, like water (H2O) and mercury (Hg).
• We have water column units for different density at temperatures, like 0 °C, 4 °C, 60 °F and 68 °F and for theoretical densities. Atmospheric units
Cgs unit of pressure
Different cgs mechanical units are derived from using these cgs base units.
more existing…
Just to mention, for case in aBeamex MC6 calibrator, there are over 40 dissimilar pressure units, plus notwithstanding a few custom units for the thrill-seekers.

Beamex force per unit area calibration tools

Source: https://blog.beamex.com/pressure-units-and-pressure-unit-conversion
0 Response to "Pressure Is Read 75.5 Cm of Mercury"
Post a Comment